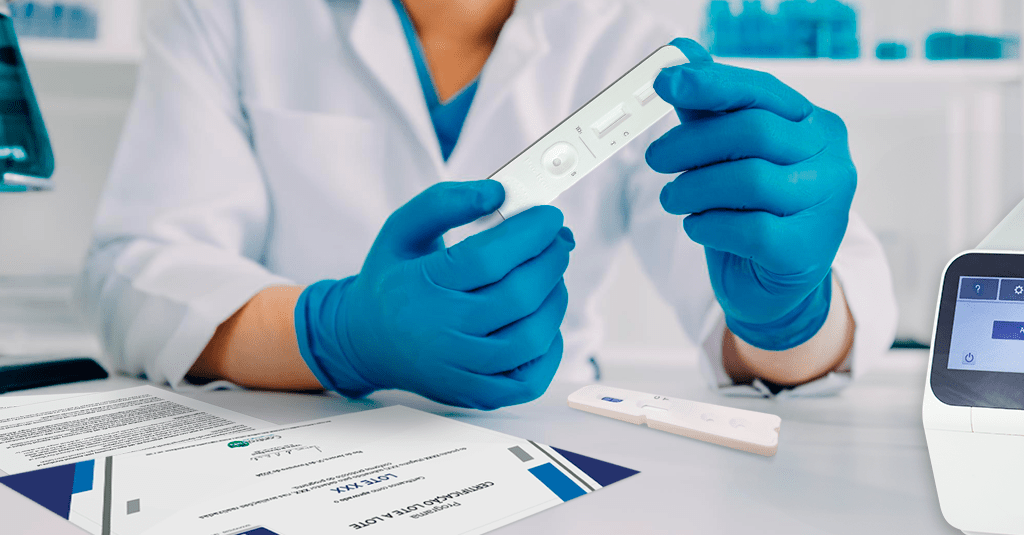
Initiative promotes increased reliability for analytical routines and greater safety for patients.
Clinical analysis establishments need to implement a quality assurance program for their results to manage risks and ensure patient safety.
These guidelines are essential to prevent deviations in reports and to promote the credibility of the establishment with the public. They are defined by government regulations, such as RDC 786/2023, or by laboratory accreditation standards such as ISO 15189, PALC-SBPC/ML, DICQ-SBAC, among others.
The pursuit of excellence underscores the importance of quality control practices in maintaining rigorous standards. Within these practices, it is essential to ensure that the replacement of reagent batches does not affect analytical routines, as recommended by national and international reference sources such as CLSI EP26.
Innovative program
Controllab, a reference in solutions for quality control of analyses, offers the Lot-to-Lot Certification Program for POCT device manufacturers with visual and automated reading.
In this program, batches of these products are evaluated according to the main guidelines and related literature, using representative materials from routine. The goal is to verify if the results obtained between batches are consistent and maintain analytical performance.
Manufacturers that submit their devices to the program meet the needs of laboratories, pharmacies, clinics, and offices, helping them reduce costs and efforts in monitoring the replacement of new reagents in the analytical routine, promoting:
- risk reduction;
- greater patient safety;
- confidence in the product.
Furthermore, manufacturers demonstrate quality to the market and assist clinical analysis establishments in evidencing compliance with regulatory and legal guidelines.
In this scenario, Grupo Kovalent, committed to quality requirements and its audience, is at the forefront in ensuring certification for a selection of its products. The following batches are already certified:
The Lot-to-Lot Certification Program is an innovative initiative for the segment in Brazil. This pioneering spirit is a hallmark of Controllab, known for competence, experience, trust, and innovation. The company is recognized by the main standards related to its operations: ISO 9001, 17025, 17034, and 17043.
For more information about the Lot-to-Lot Certification Program and other solutions for quality control of analyses, contact Controllab.