Background
Laboratory Accreditation Programs play a crucial role in ensuring that laboratories (labs) adhere to quality management systems based on best practices, meeting stakeholders’ key requirements. To assess their processes, organizations employ performance monitoring systems, often relying on performance indicators (IND). In Brazil, the Metricare Indicators platform (Metricare), facilitates the comparison of IND for labs performance across dimensions and aims to help labs identify improvement opportunities and enhance overall market performance.
Aim
This study aims to compare laboratory performance metrics within the indicator’s platform, focusing on the impact of laboratory accreditation.
Methods
This study, conducted from Jan/22 to Jul/23, focused on 6 essential INDs: Proficiency Test Performance (PTP), Incorrect Reports (IR), Failure to Communicate Critical Results (FCCR), Delay in Outpatient Results (DOR), Sample Recollection (SR), and Patient Satisfaction (NPS). The IND, aligned with International Federation of Clinical Chemistry (IFCC) harmonization, were submitted monthly by participating labs to Metricare. The performance
evolution analysis involved comparing group medians for each IND in a global sample. Labs were stratified in groups based on accreditation status: (A) Accredited by the Brazilian Society of Clinical Pathology and Laboratory Medicine (SBPC/ML); (B) Accredited by other programs; (C) Non-accredited labs. Sigma (s) metrics were used for all IND, except for the patient satisfaction IND, expressed as a percentage.
Results
Two IND (IR, s 5.2; SR, s 3.9) performed similarly across all 3 groups. The remaining 4 IND showcased superior performance by group A. For instance, in the PTP IND, group A labs exhibited s 3.7, outperforming groups B (s 3.6) and C (s 3.4). The pattern persisted across other IND, including DOR (groups A, B, and C s 3.7, 3.5, and 2.7 respectively), FCCR (groups A, B, and C s 4.6, 3.4, and 4.0 respectively), and NPS (groups A, B, and C percentages, 91%, 88%, and 74%, respectively) (Table 1).
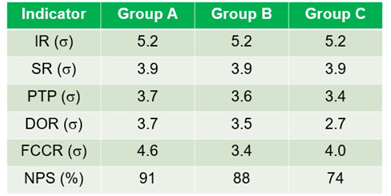
Incorrect Reports (IR); Sample Recollection (SR); Proficiency Test Performance (PTP); Delay in Outpatient Results (DOR); Failure to Communicate Critical Results (FCCR); Patient Satisfaction (NPS); Group A (Labs Accredited by the Brazilian Society of Clinical Pathology and Laboratory Medicine (SBPC/ML); Group B (Labs Accredited by other programs); Group C (Non-accredited Labs).
Conclusions
This study underscores the positive impact of laboratory accreditation programs on organizational performance,
benefiting patients and stakeholders. Notably, differences in performance among labs accredited by different programs highlight the importance of careful program selection. Labs should consider these findings when choosing accreditation programs to ensure optimal outcomes and overall excellence.
References
Shcolnik W, Berlitz F, Galoro C A O, Biasoli V, Lopes R, Jerônimo D, Balli L B, Bernardes L. Brazilian laboratory indicators benchmarking program: three-year experience on pre-analytical quality indicators. Diag 2021; 8: 257-268.
Sciacovelli, L., & Plebani, M. (2009). The IFCC Working Group on laboratory errors and patient safety. Clinica chimica acta; international journal of clinical chemistry, 404(1), 79–85.
Panhwar A., Naeem M.A., Haq A.U., Zainulibad S., Ahmad M., Haq S.U. Laboratory management system and competency of accredited laboratories. Int. Rev. Basic Appl. Sci. 2020;8(2):9–13.
