See where your organization stands today and how far it can go with analytics-driven quality insights.
Quality management in healthcare has evolved. Today, it is not enough to simply monitor results; they must be interpreted with analytical intelligence, performance compared, and opportunities for improvement identified quickly and accurately.
With this in mind, Controllab presents Analytical QC BI, the new module of Metricare that transforms Proficiency Testing data (External Quality Control) into strategic insights for comparative management (benchmarking).
With it, laboratory and healthcare network managers can see beyond the numbers: they discover where their units excel, where improvements are needed, and how to align their practices with the best industry standards.
Clarity and confidence for decisions that drive results
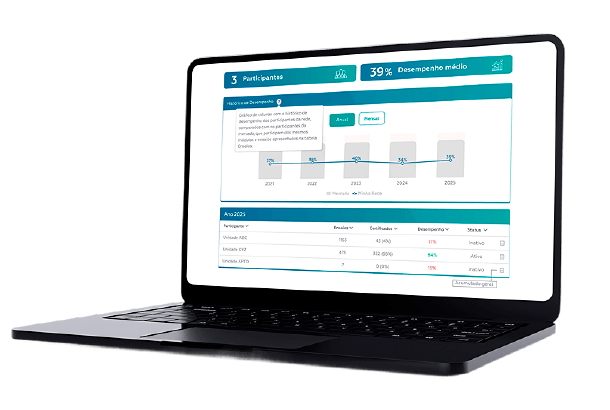
Analytical QC BI automatically organizes Proficiency Testing data, enabling comparisons between laboratories within the same network and with the market.
This broader perspective strengthens governance, enhances traceability, and facilitates performance demonstration in audits and accreditations.
With consistent and up-to-date information, managers gain clarity to act, lead their teams with greater confidence, and turn data into decisions that elevate organizational performance.
At the 57th CBPC/ML, the Rede D’Or case was presented, highlighting how they already use Analytical QC BI as an ally in data-driven quality management, consolidating their position as a reference in innovation and analytical intelligence in healthcare.
Security and reliability
The data processed in Metricare – the intelligence platform that supports daily decision-making in healthcare and wellness organizations – is secure, confidential, and anonymized. The tool gathers information on laboratory management, microbiology, patient safety, and other areas, providing a comprehensive view of performance. Check the full list.
The platform also integrates systems from technology partner companies, facilitating data collection and reducing errors.
A new level for quality management
With Analytical QC BI, your organization can:
- Compare results between units and with the market;
- Set realistic goals and track progress;
- Identify trends and respond quickly to deviations;
- Demonstrate compliance with accreditations and regulations, including the Brazilian standard RDC 978/2025 from Anvisa.
Recognition and partnerships that reinforce credibility
Discover the power of Analytical QC BI
See where your organization stands today and how far it can go with analytics-driven quality insights.
Talk to the specialists behind this innovation via WhatsApp +55 (21) 98258-0074.